IVF's Next Frontier: Breakthroughs in Embryo Selection!
Blastocyst Assessment Improves Pregnancy Outcomes and is Augmented by Innovative Machine Learning Techniques
Introduction:
In Vitro Fertilization (IVF) has revolutionized reproduction, offering hope to countless couples facing fertility challenges. Since the birth of the first IVF baby in 1978, this technology has become a pivotal solution for those struggling with infertility and has also opened doors for people in nontraditional relationships to start families. Today, over 10 million babies worldwide have been born through IVF, a testament to its profound societal impact (Hart et al., 2022).
However, despite its successes, IVF is a relatively young field with evolving technologies and methodologies. Its effectiveness varies significantly, particularly influenced by factors such as the patient's age. For example, women under the age of 35 see a success rate of approximately 36% (live births per embryo implantation), which sharply decreases with age (Table 1). Such statistics highlight the need for ongoing advancements to enhance IVF's efficacy.
This article delves into some recent breakthroughs in IVF, particularly focusing on those that have the potential to not just improve birth rates, but also to make the process more efficient, cost-effective, and accessible. We will explore how recent studies shed light on the intricate details of embryo quality and the promise of non-invasive techniques in predicting embryo viability, painting a picture of a future where IVF is more successful and patient-centric.

Overview: How does IVF work?
To set the groundwork for the advancements in IVF, let’s first walk through the IVF process.
Ovarian Stimulation: IVF treatment commences with ovarian stimulation. Women are born with all the eggs in their lifetime - they do not produce new ones. Instead of maturing and releasing one egg per menstrual cycle, the IVF drugs stimulate the ovaries to mature and release multiple eggs for harvesting.
Egg Retrieval: Once the eggs are mature, a precisely timed procedure called follicular aspiration is performed. Under mild anesthesia, a thin needle is inserted through the vaginal wall to extract eggs from the ovaries.
Fertilization and Embryo Culture: After egg retrieval, the mature eggs are combined with sperm in a laboratory setting to facilitate fertilization. Post-fertilization, the embryos are carefully cultured in a controlled environment. This crucial phase typically lasts about 5 to 6 days, during which the embryos develop into blastocysts (Figure 1). Keep in mind, even at the blastocyst stage, an embryo is still significantly smaller than the average grain of sand (which is approximately 0.5 mm)! A time-lapse video (Video 1) depicts this incredible journey from fertilization to the blastocyst stage.
You may be asking why this stage is critical. Blastocysts are more developed and have a higher chance of implantation once transferred to the uterus. At this advanced stage, they have distinct cell types and a fluid-filled cavity, making them more resilient and capable of successful attachment to the uterine lining. This key developmental milestone is when embryologists can best assess the embryos' viability, selecting those with the highest potential for a successful pregnancy.
Embryo Transfer: The final step involves transferring one or more selected embryos into the uterus. This decision is critical and is influenced by the embryos' development stage and quality. The transfer is done using a catheter through the cervix into the uterus. Post-transfer, supplemental progesterone is often prescribed to enhance the uterine lining's receptivity.
Follow-up and Freezing: If the embryo implants are successful, pregnancy can occur. Any additional healthy embryos can be cryopreserved for future use.

Video 1. A fertilized embryo developing from a single cell to a blastocyst. This is a time lapse video that takes place over five days. Credit to Boston IVF.
Claims:
1. Blastocyst quality is a predictor of live birth rates in IVF:
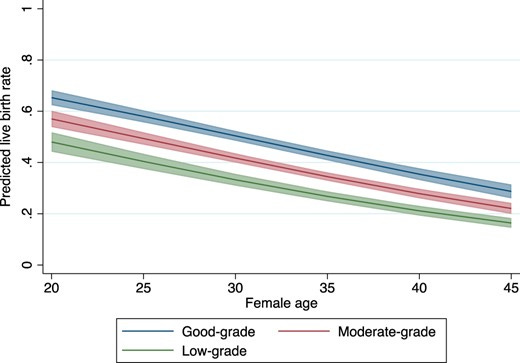
As mentioned above, embryos are implanted for IVF at the blastocyst stage (approximately five days post-fertilization). For these procedures, selecting the best blastocyst is believed to play a crucial role in increasing the success of embryo implantation and resulting pregnancy. However, the extent to which embryo quality impacts this has not been thoroughly established. Now, a recent study by Zou et al. (2023) provides significant insights into this aspect. Their multinational, multicenter observational study analyzed over 10,000 single blastocyst transfer cycles and categorized blastocysts into good, moderate, and low grades based on morphology (morphology are the visual characteristics of the embryo).
The study found that while good-grade blastocysts yielded a live birth rate of 44.4%, moderate and low-grade blastocysts had lower rates at 38.6% and 30.2%, respectively (Figure 2). Notably, even very low-grade blastocysts (CC grade) still had a reasonable chance of success, with a 13.7% live birth rate. These findings suggest that while higher-quality blastocysts improve the likelihood of live birth, even lower-grade embryos can result in successful pregnancies. This information could be vital for patients with limited high-quality embryos or those who have experienced repeated implantation failures.
2. Blastocyst health can be predicted from embryo culture media
You may have left the previous section wondering what constitutes a good embryo morphology or how clinicians even score an embryo. Currently, the assessment of these embryos is performed morphologically, that is, looking at the visual features of the embryo under a microscope. This may seem less than the robust standard you would want for such a critical decision. However, some of this appears to be changing!

A study by Wu et al. (2024) presents a groundbreaking non-invasive technique to assess blastocyst health by analyzing the liquid medium in which the embryo develops, as illustrated in Figure 3. In the study, the team employed a machine-learning model to evaluate embryo quality. This innovative approach involved analyzing the levels of extracellular RNA (exRNA) in the culture media. The model was trained to identify patterns in the exRNA data that correlate with the health and viability of blastocysts. By applying this machine-learning algorithm, the study could predict the quality of embryos based on the molecular signatures found in the culture media, offering a sophisticated, non-invasive tool for improving embryo selection in IVF procedures.
Impact:
The recent advancements in IVF, particularly in blastocyst health assessment and selection, have significant implications:
IVF Efficiency Improvements: The correlation between blastocyst quality and pregnancy success emphasizes the need for precise embryo grading. These findings could pave the way for more effective IVF treatments, potentially increasing success rates per cycle and reducing the number of cycles needed, which is a significant benefit for patients both emotionally and physically.
Cost Reduction: Currently, IVF treatments are approximately $15,000 per cycle, with most women requiring multiple cycles. Improvements to embryo selection efficiency could reduce costs, benefiting individuals and healthcare systems.
Health Implications for IVF Children: The long-term health effects on children born through IVF are a subject of ongoing investigation. Research has identified various health impacts in IVF-born children, including elevated blood pressure, advanced bone age, and increased BMI (Hart et al., 2013, Wolff et al., 2022, Laugesen et al., 2023) (Table 2). The exact causes of these risks—whether they stem from the IVF procedures themselves, genetic predispositions, or specific obstetric outcomes—are not yet fully understood. However, the potential role of embryo quality in these health outcomes is an interesting aspect that warrants further investigation. Could selecting higher-quality embryos, for instance, lead to a reduction in these risks? This question highlights the importance of continued research in this area.

Skeptical Analysis of Recent IVF Research:
While these studies offer promising advancements in IVF technology, several considerations warrant a cautious approach:
Morphological Assessment Limitations: The reliance on morphological assessments raises questions about subjectivity in grading blastocyst quality. Morphological criteria might not fully capture the complexities of embryo viability.
Machine Learning Model Validity: The use of machine learning to analyze exRNA profiles is innovative but requires rigorous validation. The model's accuracy in different clinical settings and its adaptability to varied patient profiles need thorough evaluation.
Limited Predictive Power of Blastocyst Quality Score: This suggests that while blastocyst quality indicates potential success, it is not the sole determinant. Other factors like genetic quality, maternal health, and uterine conditions are crucial and often not captured by morphological scores alone.
Conclusions and Next Steps:
The recent advancements in IVF technology, particularly in understanding embryo viability and selection, highlight the necessity for a more integrated and multifaceted approach in future research. This approach should amalgamate current methods like morphological evaluation with advanced techniques such as genetic testing and exRNA profiling. Moreover, incorporating a wider range of factors—such as maternal health, sperm quality, and lifestyle factors—is crucial to enhance the personalization and effectiveness of IVF treatments. Moving forward, the focus should be on developing comprehensive, multi-factorial models that can accurately predict and improve IVF success rates, ensuring that these scientific advancements translate into tangible benefits for patients.
Support:
These newsletters take a significant amount of effort to put together and are totally for the benefit of the reader. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
Society for Assisted Reproductive Technology (SART). Clinic Summary Report. Retrieved from SART (Accessed 20 January 2024).
ESHRE ART fact sheet (2022). Available at: https://www.eshreeu/Europe/Factsheets-and-infographics (Accessed 20 January 2024)
Brison, D.R., 2022. IVF children and healthy aging. Nature Medicine, 28(12), pp.2476-2477.
Firmin, J. and Maître, J.L., 2021, December. Morphogenesis of the human preimplantation embryo: bringing mechanics to the clinics. In Seminars in Cell & Developmental Biology (Vol. 120, pp. 22-31). Academic Press.
Hart, R. and Norman, R.J., 2013. The longer-term health outcomes for children born as a result of IVF treatment: Part I–General health outcomes. Human reproduction update, 19(3), pp.232-243.
Hart, R.J. and Wijs, L.A., 2022. The longer-term effects of IVF on offspring from childhood to adolescence. Frontiers in Reproductive Health, 4, p.1045762.
Laugesen, K., Veres, K., Hernandez-Diaz, S., Chiu, Y.H., Oberg, A.S., Hsu, J., Rinaudo, P., Spaan, M., van Leeuwen, F. and Sørensen, H.T., 2023. Overweight or obesity in children born after assisted reproductive technologies in Denmark: A population-based cohort study. Plos Medicine, 20(12), p.e1004324.
Von Wolff, M. and Haaf, T., 2020. In Vitro fertilization technology and child health: risks, mechanisms and possible consequences. Deutsches Ärzteblatt International, 117(3), p.23.
Wu, Q., Zhou, Z., Yan, Z., Connel, M., Garzo, G., Yeo, A., Zhang, W., Su, I., Zhong, S., 2024. A temporal extracellular transcriptome atlas of human pre-implantation development. Cell Genomics, 4(1).
Zou, H., Kemper, J.M., Hammond, E.R., Xu, F., Liu, G., Xue, L., Bai, X., Liao, H., Xue, S., Zhao, S. and Xia, L., 2023. Blastocyst quality and reproductive and perinatal outcomes: a multinational multicentre observational study. Human Reproduction, 38(12), pp.2391-2399.



Superb analysis, David. Thank you for all of this information and your grounded analysis.
It's such a large drop in live birth rates post 40 years old in Table 1.
I love these articles. They're so well done: both a thorough discussion of the technology and accessible to non-scientists.
I've been trying to figure out how to upgrade to a paid subscription, but on the "you're subscribed" page under "status" it just says "free subscriber," and there's no option for doing anything else. Am I doing it wrong?