Foreword
There is a growing interest in human longevity, encompassing both the extension of healthspan and lifespan. This fascination is evident in the endeavors of public figures, which range from tech entrepreneurs like Bryan Johnson to scientists like Harvard’s David Sinclair, who famously stated, ‘The first 150-year old has already been born’. While human life extension breakthroughs are still on the horizon, significant progress is being made in animal longevity, particularly in dogs. This is perhaps better grounds for proof-of-concept work in longevity because of the shorter lifespan of dogs and potentially easier aging-related bottlenecks to solve. Most importantly, though, these therapies may hold promise for human longevity as well.
This article delves into the pioneering products from Loyal, which have the stated purpose of extending canine longevity. We will explore the first-of-its-kind drug, LOY, its scientific basis, its potential impact, and the ethical considerations it raises, offering insights into how this venture in veterinary medicine might foreshadow advancements in human health and aging.
Introduction:
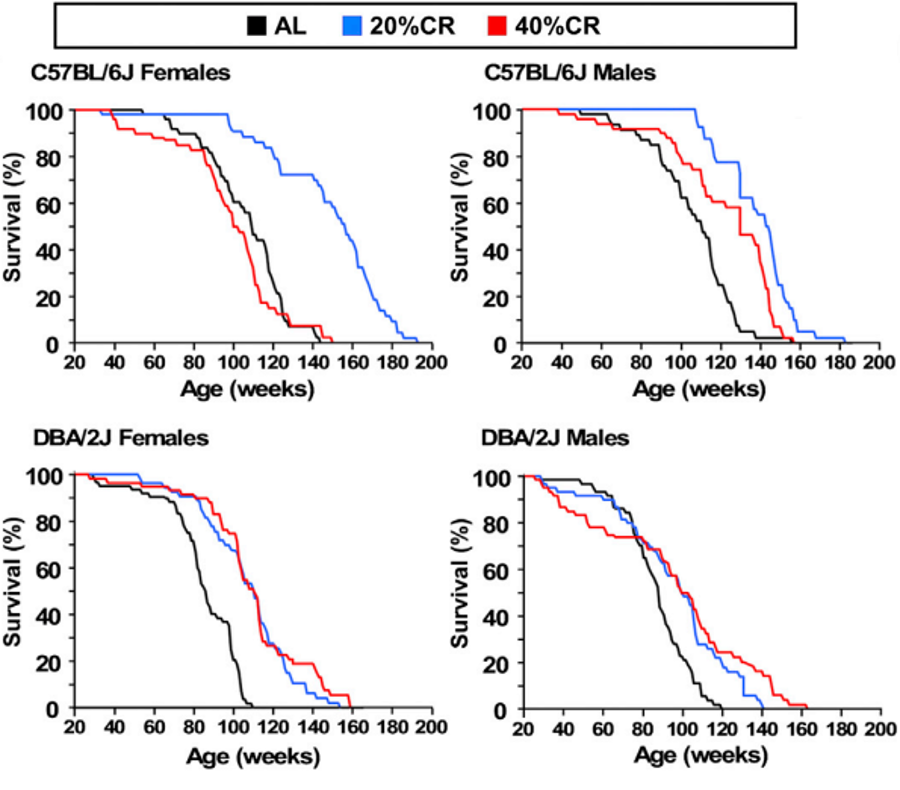
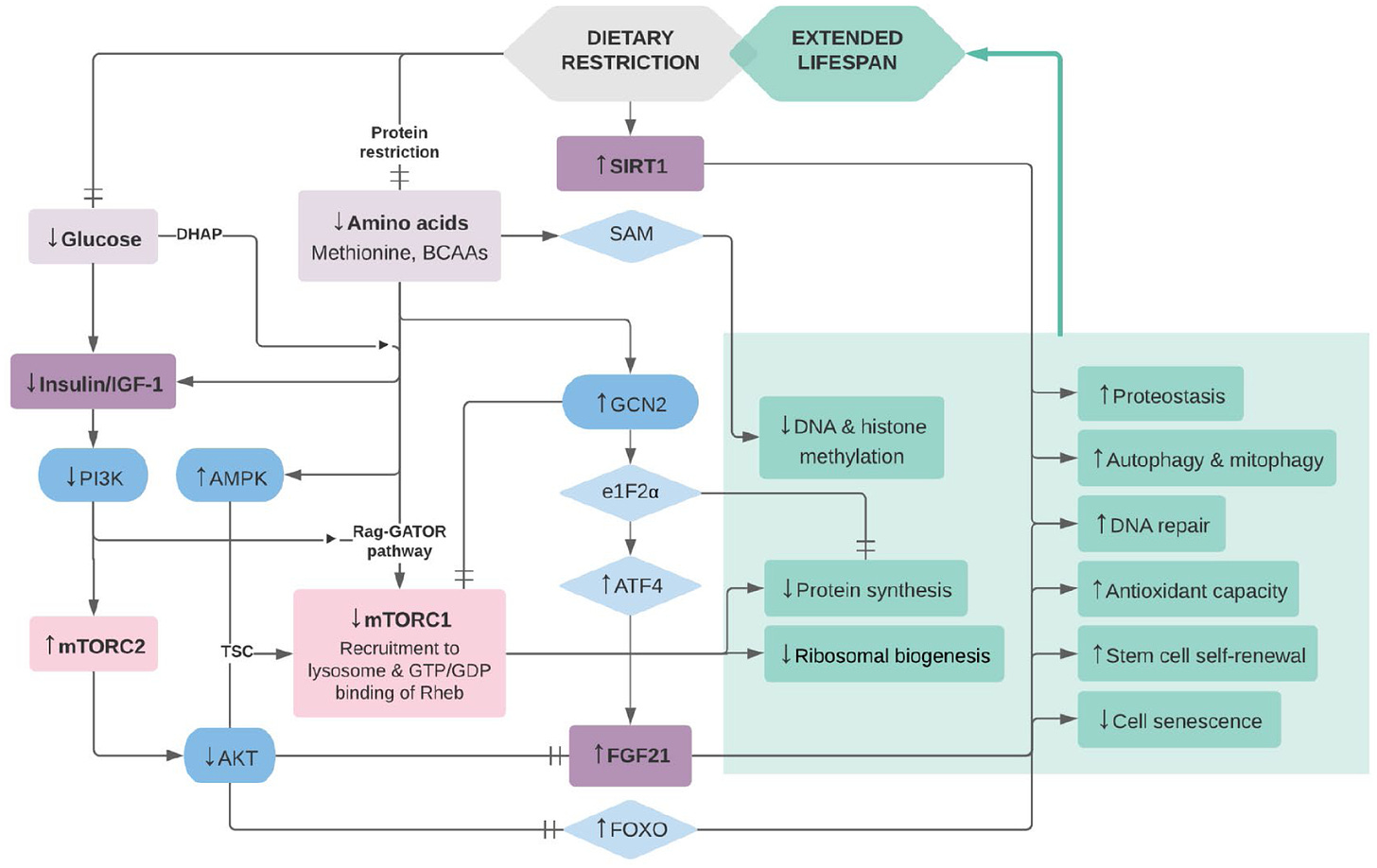
What causes aging on both a cellular and holistic level is not fully understood. It is believed to be a multifactorial challenge, encompassing factors like genetics, epigenetics, cellular senescence, stem cell depletion, and others. However, we have learned a significant amount about life extension in model organisms that appear to be translatable in larger mammalian organisms, including humans (Green et al, 2021). One repeatedly observed phenomenon is dietary restriction (between 10-50%) without malnutrition, appears to enhance lifespan significantly, as shown in Figure 1 ( NOTE: it would have been interesting if Figure 1 opted to include a group with a caloric excess to see if this reduced % survival vs time). While the mechanism of caloric restriction on longevity is not fully understood, there is a clear connection to metabolism and hormones, particularly growth hormone (GH) and insulin-like growth factor (IGF). This may be bad news news for bodybuilders, athletes, and generally, people who enjoy eating (including myself).
Okay, thanks for the biology class, David. What does this have to do with my dog living longer?
Well, remember how I mentioned IGF is a regulator of aging? Some clever researchers noticed two things:
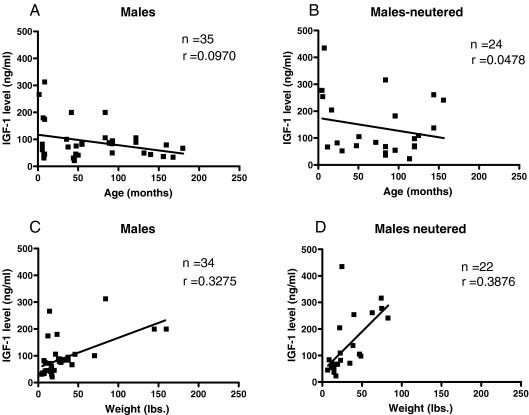
Large dogs live much shorter lives than small dogs (Figure 2). In fact, lifespan and size are inversely related.
The blood concentration of IGF is disproportionately higher in large dogs compared to small dogs (Figure 3). This seems to be proportionally correlated.
Cue Loyal, a San Francisco-based biotech company, with its groundbreaking development of LOY, an anti-aging drug specifically designed for canines. Loyal's LOY targets IGF-1, reducing it in large dog breeds. Moving forward, we will discuss their claims, path to commercialization, the impact of this on longevity, and next steps.

Claim(s):

At the heart of Loyal's innovative endeavor is its anti-aging drug, LOY, which presents a bold claim in the field of veterinary medicine. The drug's primary target is the Insulin-like Growth Factor-1 (IGF-1) hormone, a key player in cell growth and metabolism. Research has shown that IGF-1 serum concentrations are significantly higher in larger dog breeds compared to their smaller counterparts (Figure 3). This disparity in hormone levels is believed to be a contributing factor to the accelerated aging process observed in larger breeds, often resulting in a considerably shorter lifespan.
LOY claims to reduce the levels of IGF-1 in larger dogs, bringing it down to levels observed in smaller breeds. This reduction aims to slow down the aging process, thereby potentially extending the healthy lifespan of these dogs. LOY-001, Loyal’s flagship drug, was tested in an FDA-accepted model of canine aging study (n=451 dogs). Most importantly, the results claimed an improvement in clinically relevant aging parameters.
On Loyal’s website, there appear to be several formulations of this drug with slightly different indications and formulations (Table 1). The most advanced formulation, LOY-002, is an oral form given daily and is currently being tested in a clinical trial. This target population is older dogs with all sizes larger than the smallest dogs (the website indicates this is dogs less than 15 lbs).
Mechanism:
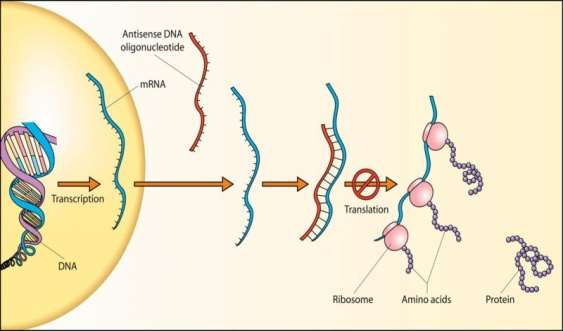
I had difficulty finding out exactly ‘how’ Loyal reduces IGF-1 in dogs. The best I could find came from a Wikipedia source. This describes the approach as an RNA antisense treatment. Antisense RNA is a form of therapy that involves using RNA molecules to interfere with the expression of specific genes (Figure 4). In the case of LOY-001, this approach likely targets the gene(s) involved in IGF-1 production or regulation, thereby reducing the hormone's levels in the body. This method differs from other approaches like monoclonal antibodies or small molecule inhibitors, which typically target proteins or pathways after they have been produced. It is important to note that this information comes from a secondary source!
Impact:
The potential impact of LOY-001 extends far beyond the realm of veterinary medicine, offering a glimpse into a future where the aging process in pets, and possibly even in humans, can be managed more effectively. This drug represents a monumental stride in canine healthcare, especially for larger breeds that historically have faced a shorter lifespan due to accelerated aging processes.
For Canine Health and Longevity:
Extended Lifespan for Larger Breeds: By targeting the IGF-1 hormone, LOY could significantly increase the lifespan of larger dog breeds, allowing them to enjoy more years of vitality and health. This is particularly impactful for breeds that traditionally live shorter lives due to their size.
Improved Quality of Life: The prospect of extending a dog's life carries the profound benefit of enhancing the quality of time pet owners spend with their companions. Healthier, longer-lived pets mean more years of companionship, love, and shared experiences.
Broader Implications in Anti-Aging Research:
Model for Human Longevity Studies: Dogs share many age-related ailments with humans and experience them within a condensed timeframe. This makes them ideal models for studying aging and longevity. Success with LOY could inform similar approaches in human anti-aging treatments.
Advancement of genetic medicine: The therapy (if utilizing an antisense RNA strategy) would indeed be a big advancement for this type of procedure.
A Paradigm Shift in Veterinary Medicine:
Innovative Treatment Approach: LOY-001's method of targeting a specific hormone to extend lifespan is a novel approach in veterinary medicine. It sets the stage for future research and development of treatments that can tackle age-related issues more precisely.
Preventive Health Care: The drug could also mark a shift towards more preventive health care in pets, addressing age-related issues before they manifest severely, thereby enhancing overall pet health standards.
In essence, the impact of LOY lies in its potential to fundamentally alter our understanding of aging in animals, opening up new avenues for research and treatment. It symbolizes a leap towards a future where the longevity of our pets is no longer left to fate but can be enhanced through scientific breakthroughs.
Skeptical Analysis and Questions:
Now that we’ve hyped this sufficiently, let’s pull back and have some skepticism for this product along with some thoughtful questions!
Lack of transparency: Thus far, I have been unable to find key data demonstrating Loyal’s efficacy with the LOY drugs. One would expect simple literature demonstrating the drug mechanism of action and proof of efficacy in an in vitro model or preclinical studies. Until we see some of this data, I will remain highly skeptical. In addition, there appear to be three different products in various stages of testing: LOY-001, LOY-002, and LOY-003. They appear to have different target groups, but it’s unclear if this is just different dosing strategies (oral vs injection) for the indications or fundamentally a different drug.
Safety and other effects of IGF-1 knockdown: It’s unclear if there are safety implications of knocking down IGF-1 in dogs. It would be interesting to know why IGF is so elevated in large dogs in the first place. What may be the tradeoff for knocking it down?
Timing for IGF inhibition: Is starting dosing of IGF inhibitors late in life too little too late? I always thought of aging as an accumulated effect and performing this work in the final expected years of life, I would imagine, only observing marginal life extension. However, it is clear that this is among the indications being assessed “LOY-002 is being developed to support healthy aging in older dogs of nearly every size and breed.”
Efficacy expectation: There are several authors that make the claim IGF is not the key regulator of aging and there is a greater implication for growth hormones (Brown-borg, 2022). It will be interesting to see the signal from these clinical studies.
Ethical considerations: I think there are some fairly open-ended questions here, and I’ll list a few that I can think of. What is an appropriate amount of money to spend on a pet’s medical bills? How long should we extend the life of animals (or potentially people someday)? If a magic injection is available to double your pet’s life and health span, is it ethical to allow it to age naturally?
Next steps:
Loyal is taking a unique pay to approve their drug through the FDA’s Expanded Conditional Approval pathway. This pathway is meant to accelerate innovative animal drug therapies. Thus far, the FDA Center for Veterinary Medicine has approved the Reasonable Expectation of Effectiveness section for Loyal’s conditional approval application. The next step will be for the completion of the Manufacturing and Safety sections, giving the official conditional approval to market the drug for large-dog lifespan extension.
In other news, Loyal has an ongoing large-scale clinical trial for LOY-002 involving approximately 1,000 dogs. This double-blinded placebo-controlled study will give critical safety and efficacy data to support the FDA approval.
Support:
These newsletters take a significant amount of effort to put together and are totally for the benefit of the reader. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
https://en.longevitywiki.org/wiki/LOY-001
https://news.harvard.edu/gazette/story/2023/01/has-first-person-to-live-to-be-150-been-born/
Brown-Borg, H.M., 2022. Growth hormone, not IGF-1 is the key longevity regulator in mammals. The Journals of Gerontology: Series A, 77(9), pp.1719-1723.
Green, C.L., Lamming, D.W. and Fontana, L., 2022. Molecular mechanisms of dietary restriction promoting health and longevity. Nature Reviews Molecular Cell Biology, 23(1), pp.56-73.
Greer, K.A., Hughes, L.M. and Masternak, M.M., 2011. Connecting serum IGF-1, body size, and age in the domestic dog. Age, 33, pp.475-483.
Le, B.T., Raguraman, P., Kosbar, T.R., Fletcher, S., Wilton, S.D. and Veedu, R.N., 2019. Antisense oligonucleotides targeting angiogenic factors as potential cancer therapeutics. Molecular Therapy-Nucleic Acids, 14, pp.142-157.
Mitchell, S.J., Madrigal-Matute, J., Scheibye-Knudsen, M., Fang, E., Aon, M., González-Reyes, J.A., Cortassa, S., Kaushik, S., Gonzalez-Freire, M., Patel, B. and Wahl, D., 2016. Effects of sex, strain, and energy intake on hallmarks of aging in mice. Cell metabolism, 23(6), pp.1093-1112.
Selman, C., Nussey, D.H. and Monaghan, P., 2013. Ageing: it’sa dog’s life. Current Biology, 23(10), pp.R451-R453.



That's really interesting. The lack of transparency is concerning, though. It's strange that they've made it so hard to answer the questions you raised. But as a pet owner who has sworn to never have another animal because losing them is too devastating, I'd be very interested in longevity treatments if they're ultimately proven effective.
As for the ethical issues, I don't see too many in a canine application. How much someone spends on his animals is his business alone. No one else has the right to interfere with such a personal decision because no one else has the right to the pet owner's money, which is his to spend in accordance with his values. If that means improving the quality of his life by extending his pet's, no one else has the right to interfere with that decision. What's appropriate depends on the human and animal involved *in each case.* There is no fixed answer to that. Not all of the seven cats I've had over the last 23 years would have rated kidney transplants, subtotal colectomies, and femoral head ostectomies, but some have. Now that I'm retired my financial situation would result in a different decision even for the cat who got the new kidney, but just as I have no right to insist that others spend that kind of money on their pets, no one else has the right to demand that I don't spend it on mine.
Is it unethical to allow a dog to age naturally? The existence of a product isn't necessarily a moral imperative to use it. It's never been unethical to let a pet age naturally if its aging is handled with kindness, empathy, and a sense of responsibility. People recognize that allowing and prolonging an animal's discomfort, pain, and limited mobility at some point *is* unethical, and opt for euthanasia or urge the owner to opt for it.
If a pet's life can be extended without subjecting it to more age-related discomfort and pain, and if the value of its companionship is worth more to the owner than the money, then his decision to add value to his life by using something like IGF inhibitors is rational. The whole point of money is to let us both achieve our values and preserve the things that bring value to our lives.
I find it interesting how extremely tall humans also have a problem with a quicker degradation of their life spans. I'm not sure how the data correlates from humans to dogs but do they share a lot of the same reasons for their lower life spans that taller people do?