
Happy Monday Morning, Readers. Let’s be relentless this week!
Before we begin, a couple of quick housekeeping notes. It looks like we’ve gained a few new readers recently, so I want to extend a warm welcome to the Neural NeXus Newsletter! If this is your first time using Substack, feel free to explore the website—we’ve got a growing library of articles covering biotechnology, AI developments, and some fun one-off topics. I’ve linked a few popular ones below. Also, for those of you who prefer listening to reading, the Substack mobile app offers an audio feature you might enjoy. Lastly, if you find these articles valuable, consider supporting by liking, commenting, or sharing them with a friend!
Popular articles:
In this week’s BioWire, we explore a range of topics that highlight the innovation and progress driving the industry forward, such as cutting-edge therapies targeting chronic conditions like osteoarthritis and obesity, to innovative drug delivery systems for neurological disorders. We’ll also be touching on an interesting anti-aging study in dogs, which seems to be a growing trend in the burgeoning field of longevity. Let’s get started!
Ex-Pfizer Osteoarthritis Pain drug
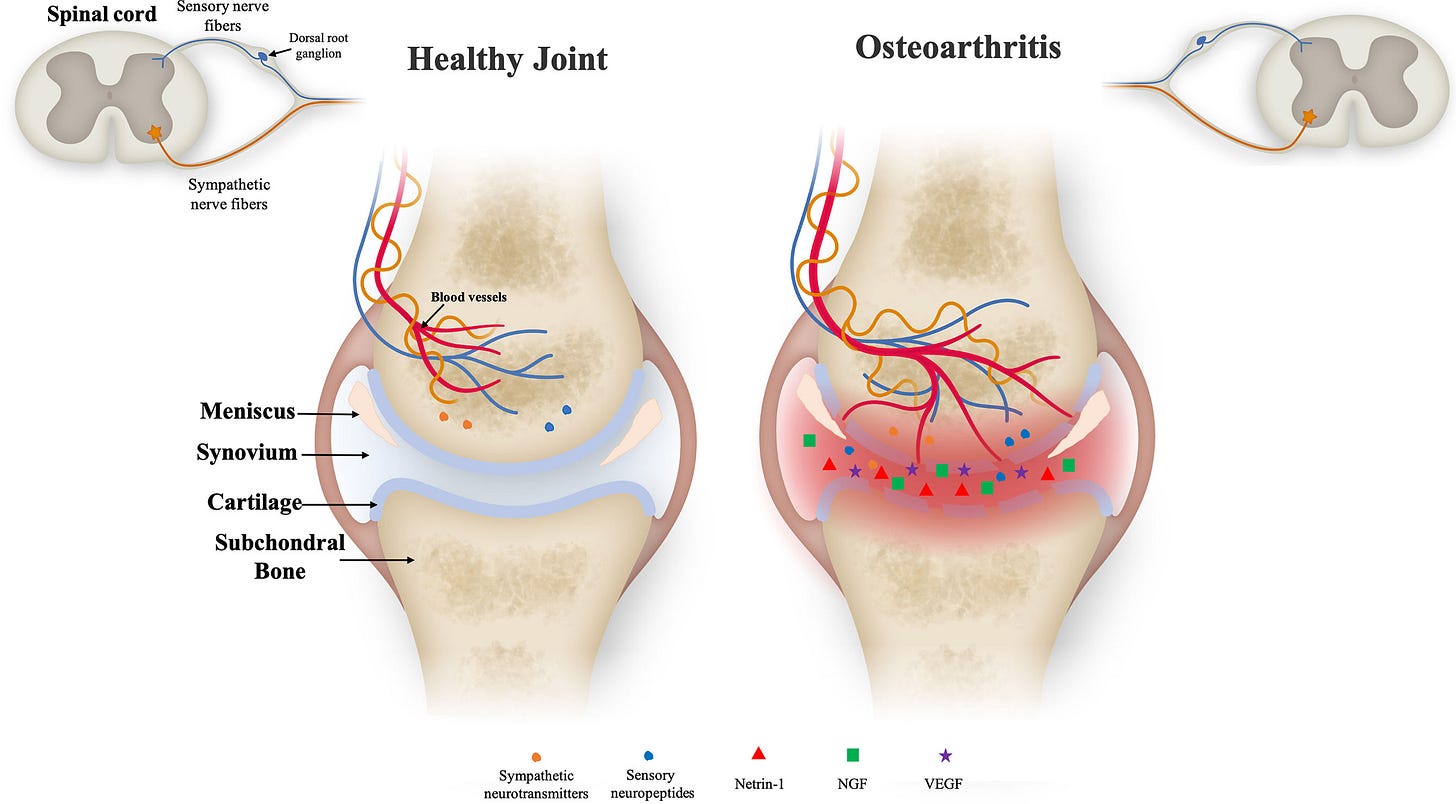
Osteoarthritis is a chronic condition characterized by the gradual breakdown of cartilage in joints, leading to pain, stiffness, and reduced mobility. This pain is often driven by inflammation and the overproduction of nerve growth factor (NGF), a protein that plays a crucial role in sensitizing nerves and amplifying pain signals. Targeting NGF has emerged as a promising strategy not only for reducing pain but also for potentially slowing the progression of osteoarthritis.
Levicept's LEVI-04 is making waves in the biotech community following positive results from its Phase 2 clinical trial. LEVI-04 is a first in class biologic therapy designed to relieve osteoarthritis pain and potentially alter the course of the disease itself. The drug is a fusion protein known as p75NTR-Fc, which targets and neutralizes NGF, effectively reducing the pain signals and inflammation associated with osteoarthritis. What sets LEVI-04 apart from conventional pain treatments is its dual action: it not only reduces pain but also promotes cartilage regeneration, offering a comprehensive benefit for patients with osteoarthritis.
Interestingly, LEVI-04 is not the first NGF inhibitor to be tested for osteoarthritis pain. Several biotechnology companies, including Pfizer, Regeneron, and Johnson & Johnson, have all attempted to produce NGF inhibitors. While these products were successful in reducing pain, they were associated with an increased risk of joint damage and accelerated osteoarthritis progression, which led to the halting and eventual abandonment of these therapies.
It remains unclear why LEVI-04 appears to be succeeding where these past products failed. The recent Phase 2 success is particularly encouraging, showing that LEVI-04 significantly reduced pain and improved joint function in patients. According to Levicept’s website, they are preparing for a larger Phase 3 definitive study. This will certainly be something to keep an eye on!
Obesity-related Weight Loss Competition: Semiglutide vs Tirzepatide

Weight-loss medications that target the hormone GLP-1, such as Wegovy and Ozempic, are becoming important tools in treating obesity and type 2 diabetes. These products help control blood glucose levels, reduce appetite, and slow down digestion, which together can lead to weight loss. While current products like Wegovy and Ozempic consist of the active ingredient semaglutide, the market is now seeing new competing products being compared to existing options to determine which is more effective.
A recent study published in JAMA Internal Medicine compared two popular weight loss drugs: tirzepatide and semaglutide. Tirzepatide, which activates both GLP-1 and another hormone receptor, showed better results than semaglutide, which only targets GLP-1 (Rodriguez et al, 2024). In the study, people taking tirzepatide were more likely to lose significant amounts of weight—5%, 10%, or even 15% of their body weight—over 12 months. On average, those on tirzepatide lost 15.3% of their body weight, compared to 8.3% for those on semaglutide.
This study, which included data from over 41,000 people, highlights tirzepatide's effectiveness in real-world settings. Both drugs were generally safe, but tirzepatide consistently led to more weight loss across different groups, including those with and without type 2 diabetes. These findings suggest that tirzepatide could be a superior product for weight loss than semiglutide.
Advancements in Drug Delivery Past the Blood-Brain-Barrier for Neurological Disorders
A recent study published in Science Translational Medicine reveals an innovative method for delivering drugs to the brain, which could significantly improve the treatment of neurological disorders (Barker et al, 2024). One of the biggest challenges in treating these conditions is the blood-brain barrier (BBB), a protective layer that tightly controls what can enter the brain. This study focuses on a new way to deliver antisense oligonucleotides (ASOs), a type of drug that works by reducing the production of specific proteins by binding to and promoting the degradation of their corresponding mRNA.
Researchers developed a special delivery system called the oligonucleotide transport vehicle (OTV). This system uses a part of an antibody that specifically targets the transferrin receptor, a protein commonly found on the cells lining the brain's blood vessels. By binding to this receptor, the vehicle can carry the drug across the BBB and into the brain. Once inside the cells, the ASO is released as the vehicle is processed, allowing it to degrade the target mRNA and reduce the levels of harmful proteins.
The study showed that this method not only allows the drug to reach the brain more effectively but also distributes it more evenly across different brain areas. This could lead to better outcomes in treating neurological diseases, which are often difficult to manage because of the BBB. The research highlights a promising new direction for delivering treatments to the brain in a way that is less invasive and more effective than current methods.
Dog longevity!
Telomir Pharmaceuticals, in collaboration with the Cleveland Clinic, is currently conducting an innovative anti-aging study in dogs, focusing on their lead product candidate, Telomir-1. Telomir-1 is a novel small molecule designed to elongate telomeres—the protective caps at the ends of chromosomes—which tend to shorten as organisms age. By modulating telomerase activity, Telomir-1 aims to reverse age-related conditions, potentially extending healthy lifespan and improving the quality of life in both animals and humans.
The ongoing study, which began in May 2024, involves geriatric Beagle dogs and is evaluating the safety and efficacy of Telomir-1 over a 63-day period. Initial results have been promising, with the treated dogs showing good tolerance to the drug. The study also aims to assess the impact of Telomir-1 on various age-related conditions such as joint damage, gait strength, and overall physical health, with plans to extend these findings to human clinical trials in the near future.
This research is not occurring in a vacuum. There are now several anti-aging studies being conducted in dogs, such as IGF inhibition by Loyal for Dogs and the use of Mytomycin in the Dog Aging Product. Together, these studies reflect a shift in the scientific community’s approach to longevity products and a growing interest in anti-aging research. The outcomes of this and other studies in dogs could have far-reaching implications for the development of treatments aimed at mitigating the effects of aging in both animals and humans.
Support:
These newsletters take a significant amount of effort to put together and are totally for the benefit of the reader. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
https://clinicaltrials.gov/study/NCT05618782
https://ir.telomirpharma.com/news-events/press-releases/detail/110/telomir-pharmaceuticals-announces-two-pivotal-milestones-for-lead-product-candidate-telomir-1
https://dogagingproject.org/project-details
Barker, S.J., Thayer, M.B., Kim, C., Tatarakis, D., Simon, M.J., Dial, R., Nilewski, L., Wells, R.C., Zhou, Y., Afetian, M. and Akkapeddi, P., 2024. Targeting the transferrin receptor to transport antisense oligonucleotides across the mammalian blood-brain barrier. Science Translational Medicine, 16(760), p.eadi2245.
Kahraman, E., Ribeiro, R., Lamghari, M. and Neto, E., 2022. Cutting-edge technologies for inflamed joints on chip: how close are we?. Frontiers in Immunology, 13, p.802440.
Rodriguez, P.J., Cartwright, B.M.G., Gratzl, S., Brar, R., Baker, C., Gluckman, T.J. and Stucky, N.L., 2024. Semaglutide vs tirzepatide for weight loss in adults with overweight or obesity. JAMA Internal Medicine.





It’s good to see that there are new drugs on the market for diabetics. However, there are specific side effects with semaglutide. Have there been any decrease in side effects with using tirzepatide? Or different ones that we should be aware of?