Happy Monday, Readers! Let’s be relentless this week.
This edition of BioWire Weekly gives briefs on significant developments in the biotech industry, including discussion on obesity combo therapies, Malaria preventative treatments, the first fetal kidney transplant, and two new cancer therapeutics. How will these advancements shape the future of medicine?
Quality Weight Loss is Pharma's Next Step to Combat Obesity with Muscle-Preserving Treatments
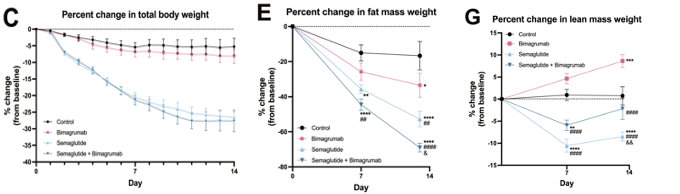
The pharmaceutical industry is exploring new strategies in the treatment of obesity. The overall approach utilizes two types of drugs that aim to enhance the quality of weight loss, particularly by preventing the loss of lean muscle alongside fat loss. This may sound familiar as we touched on it in last week’s edition.
As a reminder, current GLP-1 medications such as Novo Nordisk’s Wegovy and Eli Lilly’s Zepbound effectively reduce weight but often at the expense of muscle mass. To overcome this, biotech companies are incorporating drugs that preserve or enhance muscle development. For example, Regeneron has started a phase 2 trial to test a combination of trevogrumab with Novo's semaglutide (Wegovy) and potentially Regeneron's garetosmab, aiming to improve the quality of weight loss by preserving muscle mass. George Yancopoulos, Regeneron's Chief Scientific Officer, stated that the trial aims to not only enhance weight loss but also preserve muscle mass, which could improve metabolic health and mitigate the long-term adverse effects associated with GLP-1 usage.
Similarly, Eli Lilly is also exploring this avenue with bimagrumab, a drug designed to increase muscle mass, which they're testing in combination with semaglutide. This approach could adjust the body's composition by increasing the ratio of lean to fat mass, ideally enhancing the overall health outcomes of weight loss treatments. Both clinical and preclinical studies have shown Activin blockers such as Bimagrumab are effective at reducing fat mass while maintaining or improving muscle mass (Nunn et al., 2024; Heymsfield et al., 2021). Overall, the focus is shifting from the total weight loss to is source and proportionality.
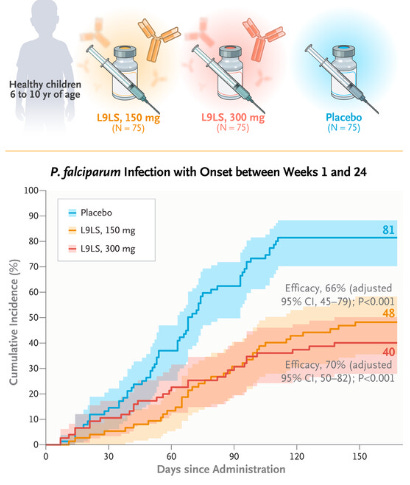
Phase 2 Trial Shows Monoclonal Antibody Reduces Malaria Incidence in Children
The study conducted in Mali explored the effectiveness of the monoclonal antibody L9LS in preventing malaria in children (Kayentao et al, 2024). The drug, administered subcutaneously, was tested over a six-month period against malaria caused by Plasmodium falciparum. The trial included two parts: 1) assessing the safety of different doses in adults and then in children, and 2) evaluating the efficacy in children compared to a placebo. Results indicated no safety concerns and showed that L9LS significantly reduced the incidence of malaria infection and clinical malaria symptoms. Specifically, children who received L9LS experienced a significantly lower risk of infection and risk of developing clinical malaria compared to those who received a placebo. This study suggests that L9LS could be an effective strategy for malaria prevention in endemic regions.
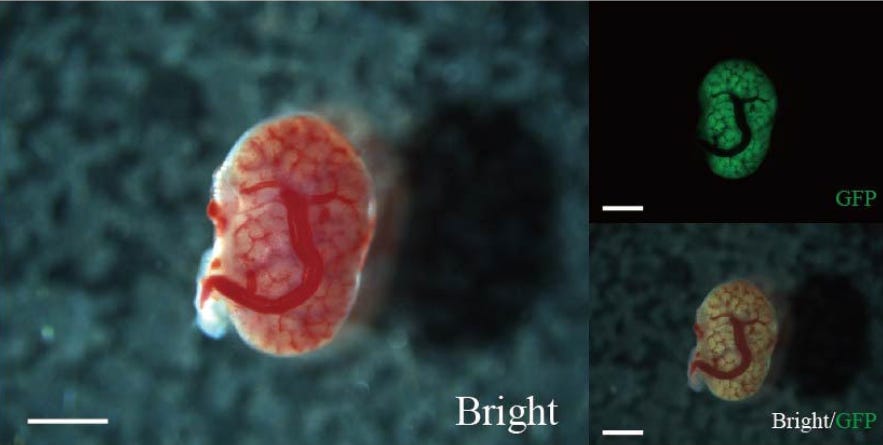
First Fetal Kidney Transplants in Rats Developed into Functioning Kidney
Surgeons have demonstrated the transplantation of kidney tissue from one rat fetus to another within the womb (Morimoto et al., 2024). Why undertake such a study? The researchers are exploring the possibility of transplanting fetal pig kidneys into human fetuses as a treatment for congenital kidney deficiencies.
In this proof-of-concept study conducted with rats, the transplanted kidneys, which were marked with green fluorescent protein for tracking, developed waste-filtering structures and began producing urine within the normal gestational period. This in-utero transplantation method shows promise in potentially reducing immune rejection, as the host's blood vessels integrated seamlessly with the transplanted tissue.
mRNA Vaccine Trial Shows Promise in Treating Aggressive Brain Cancer
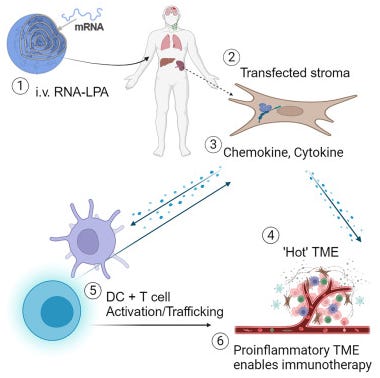
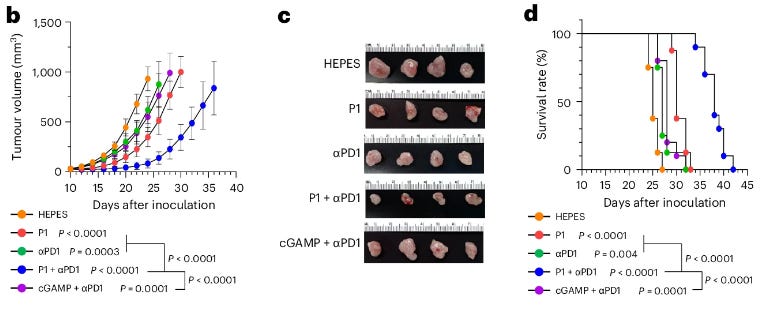
Researchers have developed a promising new mRNA vaccine aimed at treating aggressive brain cancer, tested in an innovative clinical trial that included not only human patients but also pet dogs (Mendez-Gomez et al, 2024). This vaccine was first studied in mice to model the disease and then given to 10 dogs that naturally developed brain tumors, specifically gliomas. These dogs showed extended survival times, living for a median of 139 days compared to the usual 30-60 days post-diagnosis. Encouraged by these results, the vaccine was then administered to four human patients with glioblastoma, an aggressive type of brain cancer with a typically low survival rate. Early outcomes suggest that these patients experienced extended disease-free periods or survival beyond expectations. The vaccine, personalized for each patient, works by packaging RNA from the patient's tumor into lipid nanoparticles that activate the immune system significantly. This method demonstrated effective immune responses in mice, dogs, and humans, prompting plans for an expanded Phase I trial. The study highlights the potential of this vaccine to work alongside other immunotherapies, potentially revolutionizing treatment approaches for brain cancer.
New Polypeptide-Based Immunotherapy Improves Cancer Survival in Preclinical Study
A study published in Nature Biomedical Engineering demonstrates that a new type of immunotherapy using synthetic cationic helical polypeptides effectively stimulates the immune response to combat cancer (Lee et al, 2024). Developed by scientists at the University of Texas MD Anderson Cancer Center, this novel drug leverages protein chains, or polypeptides, to significantly slow cancer growth and improve survival rates in animal models, outperforming other immunotherapy approaches especially when combined with immune checkpoint inhibitors. The polypeptides work by stimulating multiple pathways in the innate immune system—the body's first line of defense—by enhancing the immune cells' natural functions in a manner akin to the body’s own proteins. This innovative strategy not only broadens the scope of drug design but also merges synthetic design with natural biology. Author Dr. Wen Jiang highlights the potential of this approach to complement existing therapies, aiming to refine the drug’s potency and safety further to target specific cells more effectively and reduce undesirable accumulation in the liver.
Bonus: Tesla Optimus continues to impress!
We have had several articles showcasing humanoid robots (article 1, article 2). However, there has been new footage from Tesla’s Optimus, and I couldn’t resist sharing.
Support:
These newsletters take a significant amount of effort to put together and are totally for the benefit of the reader. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
Heymsfield, S.B., Coleman, L.A., Miller, R., Rooks, D.S., Laurent, D., Petricoul, O., Praestgaard, J., Swan, T., Wade, T., Perry, R.G. and Goodpaster, B.H., 2021. Effect of bimagrumab vs placebo on body fat mass among adults with type 2 diabetes and obesity: a phase 2 randomized clinical trial. JAMA network open, 4(1), pp.e2033457-e2033457.
Kayentao, K., Ongoiba, A., Preston, A.C., Healy, S.A., Hu, Z., Skinner, J., Doumbo, S., Wang, J., Cisse, H., Doumtabe, D. and Traore, A., 2024. Subcutaneous Administration of a Monoclonal Antibody to Prevent Malaria. New England Journal of Medicine, 390(17), pp.1549-1559.
Lee, D., Huntoon, K., Wang, Y., Kang, M., Lu, Y., Jeong, S.D., Link, T.M., Gallup, T.D., Qie, Y., Li, X. and Dong, S., 2024. Synthetic cationic helical polypeptides for the stimulation of antitumour innate immune pathways in antigen-presenting cells. Nature Biomedical Engineering, pp.1-18.
Mendez-Gomez, H., et al. RNA aggregates harness the danger response for potent cancer immunotherapy. Cell, (in press).
Morimoto, K., Yamanaka, S., Matsui, K., Kinoshita, Y., Inage, Y., Yamamoto, S., Koda, N., Matsumoto, N., Saito, Y., Takamura, T. and Fujimoto, T., 2024. Fetal Kidney Transplantation for In Utero Fetuses. bioRxiv, pp.2024-04.
Nunn, E., Jaiswal, N., Gavin, M., Uehara, K., Stefkovich, M., Drareni, K., Calhoun, R., Lee, M., Holman, C.D., Baur, J.A. and Seale, P., 2024. Antibody blockade of activin type II receptors preserves skeletal muscle mass and enhances fat loss during GLP-1 receptor agonism. Molecular Metabolism, 80, p.101880.



Organ transplants have to be up there with one of the coolest things our medical industry can do.
Great roundup again, David.
Really interested in the polypeptide vaccine. I did a little bit of my first postdoc on peptide vaccines for eliciting T cell responses, but this is really cool to go after innate responses using a cyclic structure.