BioWire Weekly - 018
Biotech News
Happy Monday Evening, Readers. Let’s be relentless this week!
I’m back from traveling in Thailand, and things are starting to feel back to normal. Nevertheless, there are some exciting updates to share in biotech!
Stem Cell Therapy Reverses Diabetes in Humans - A World First in iPSC Therapy
mTOR Inhibition Unlocks Dormancy in Human Blastocysts
Largest Brain Map Ever Reveals the Neural Complexity of the Fruit Fly
Microplastics Detected in the Human Brain
Stem Cells Reverse Diabetes in Humans - A World First in iPSC Therapy
Obesity and related conditions, like Type 2 diabetes, have become a constant source of discussion within BioWire. But this week, our focus shifts to a promising breakthrough for those affected by Type 1 diabetes. While nearly 40 million Americans live with diabetes—most dealing with Type 2—Type 1 patients face similar challenges in maintaining their blood sugar levels. Many of those affected are undoubtedly hoping for a cure, and lucky for them, it seems that a new wave of stem cell therapies may finally bring that hope within reach.
Induced pluripotent stem cells (iPSCs) have emerged as a promising solution in regenerative medicine. iPSCs are reprogrammed from adult cells to a stem-cell-like state, allowing them to develop into various cell types, including insulin-producing islets. The dream of researchers and clinicians has been that by creating patient-specific islets, iPSCs could help restore the body's ability to produce insulin naturally, potentially reversing diabetes rather than merely managing its symptoms. Fortunately, this appears to be on the verge of becoming a reality.
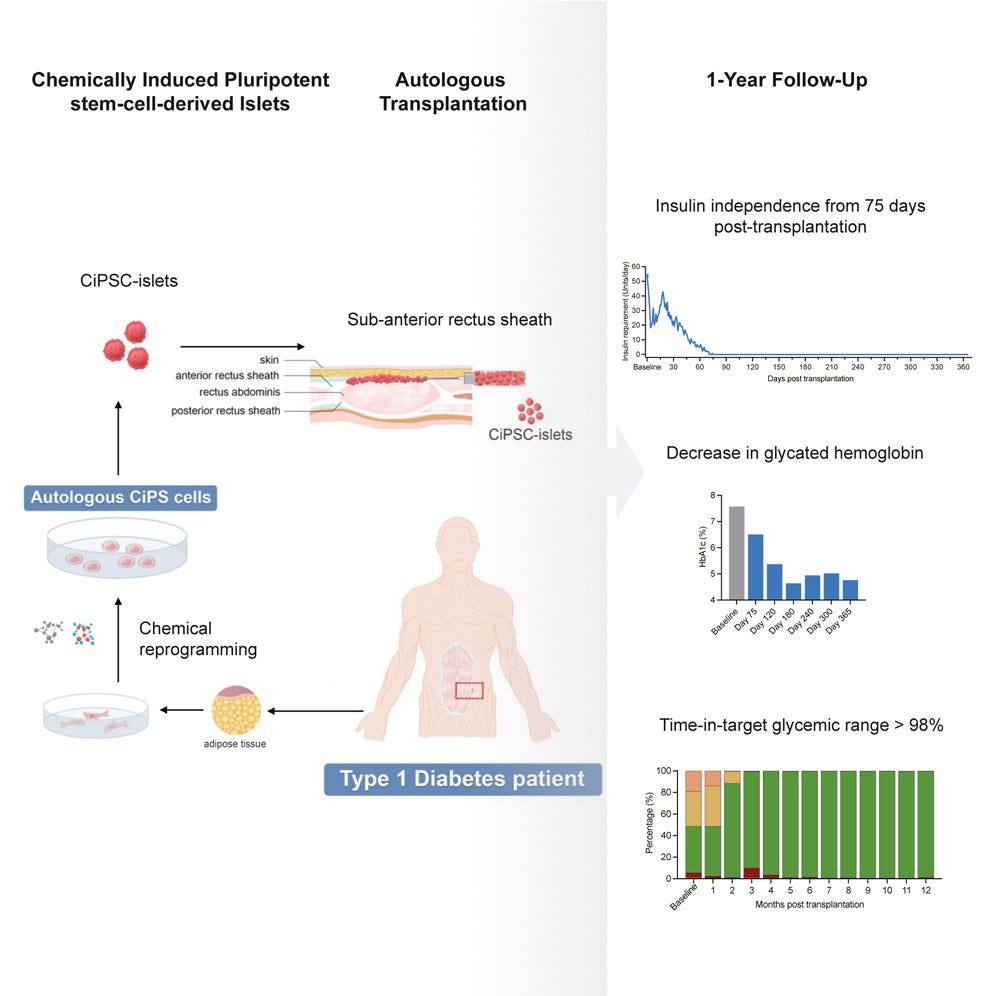
Last week, researchers reported the first 1-year results from a patient enrolled in a first-in-human phase I clinical trial assessing the use of iPSC-derived islets for type 1 diabetes (Wang et al., 2024). In this study, scientists reprogrammed cells from the patient’s abdominal fat into a stem-cell-like state, known as chemically induced pluripotent stem cells (CiPSCs). These cells were then differentiated into insulin-producing islets and transplanted beneath the abdominal anterior rectus sheath. Remarkably, just a few months after the transplantation, the patient began producing insulin and achieved stable glycemic control without the need for exogenous insulin. At the 1-year mark, the patient maintained over 98% time-in-range blood glucose levels, and the clinical endpoint was met with no observed transplant-related abnormalities.
The positive outcomes from this study suggest that CiPSC-islet transplantation could be a real path toward long-term insulin independence for type 1 diabetes patients. It’s a huge step forward in proving that CiPSCs can work in real-world treatments, not just in theory. If these cells make it into mainstream medicine, they could change how we tackle many other tough-to-treat conditions too.
mTOR Inhibition Unlocks Dormancy in Human Blastocysts
Reproductive biology and anti-aging are other hot topics at the forefront of biotechnology. This next paper combines both of these topics; a recent study published in Cell has found that turning off a specific growth pathway, called mTOR, can put early-stage human embryos into a temporary pause, similar to what happens in certain animals like mice (Iyer et al., 2024). This discovery could change the way we think about timing in human development and offer new possibilities for fertility treatments. In many animals, embryos can pause their development when conditions aren’t ideal—a state known as “diapause.” Until now, it was unclear whether human embryos could do the same.
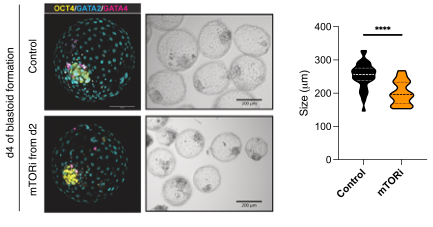
Researchers created early embryo models, called blastoids, using stem cells and used a drug to inhibit mTOR activity. This caused the blastoids to slow down and stop developing, without losing their ability to grow again later. When the drug was removed, the embryos resumed their normal growth. This means that the embryos could be paused and restarted, similar to what happens naturally in some other species.
By comparing human and mouse embryos, the researchers found that the same kinds of changes happen when the mTOR pathway is blocked, suggesting that this ability to pause might be more universal than previously thought. This new approach could help in assisted reproductive technologies, giving doctors more control over the timing of embryo development and potentially improving the chances of successful pregnancies.
Interestingly, mTOR is also a key player in aging research. Scientists have found that reducing mTOR activity can extend the lifespan of certain animals, sparking interest in its potential role in human longevity (Mannick et al., 2023). The ability to pause and restart cellular growth might not only be valuable for reproductive health but could also provide insights into how we might slow down aging processes. This connection between early development and aging underscores the broader significance of understanding mTOR's functions in human biology.
Largest Brain Map Ever Reveals the Neural Complexity of the Fruit Fly
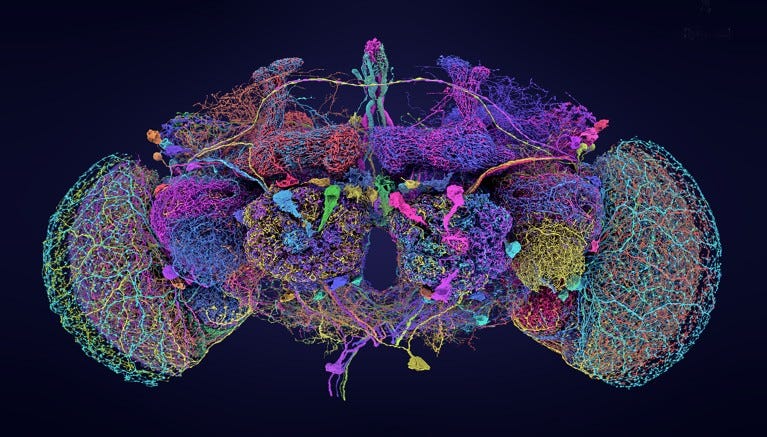
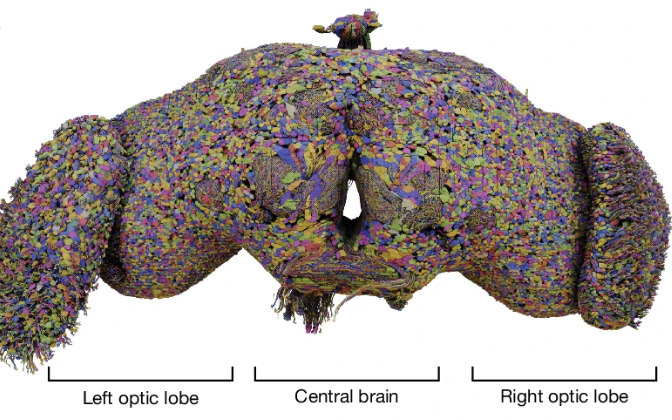
In a significant milestone for neuroscience, researchers have unveiled the most comprehensive brain map of any organism to date: a detailed wiring diagram, or connectome, of a fruit fly’s brain (Drosophila melanogaster) (Alin et al., 2024). This connectome, developed by the FlyWire consortium, captures the intricate network of nearly 140,000 neurons and over 54.5 million synapses, offering an unprecedented view into how a relatively simple brain is structured and functions. This project, published across a series of nine papers in Nature, represents a culmination of over four years of work, utilizing advanced electron microscopy and artificial intelligence tools to piece together the neural architecture.
The project wasn’t without its challenges. While AI tools played a crucial role in mapping the fruit fly brain, manual proofreading was necessary to ensure accuracy, leading to over three million manual edits. The resulting connectome revealed a staggering 8,453 types of neurons, including 4,581 that had never been previously identified. These findings underscore the fruit fly brain’s complexity and provide a valuable foundation for future research into how different types of neurons interact and function together.
Researchers have already begun exploring the potential of this map. One study used the connectome to model how sensory inputs, such as sweet or bitter tastes, trigger motor responses in the fly’s brain, demonstrating over 90% accuracy in predicting the behavior of a real fruit fly . While this map provides invaluable insights, it’s just the beginning—further studies will aim to understand electrical connectivity and develop similar maps for male flies to explore sex-specific behaviors. This achievement marks a significant step forward in our understanding of how brains, even small ones, organize and process information.
Microplastics Detected in the Human Brain
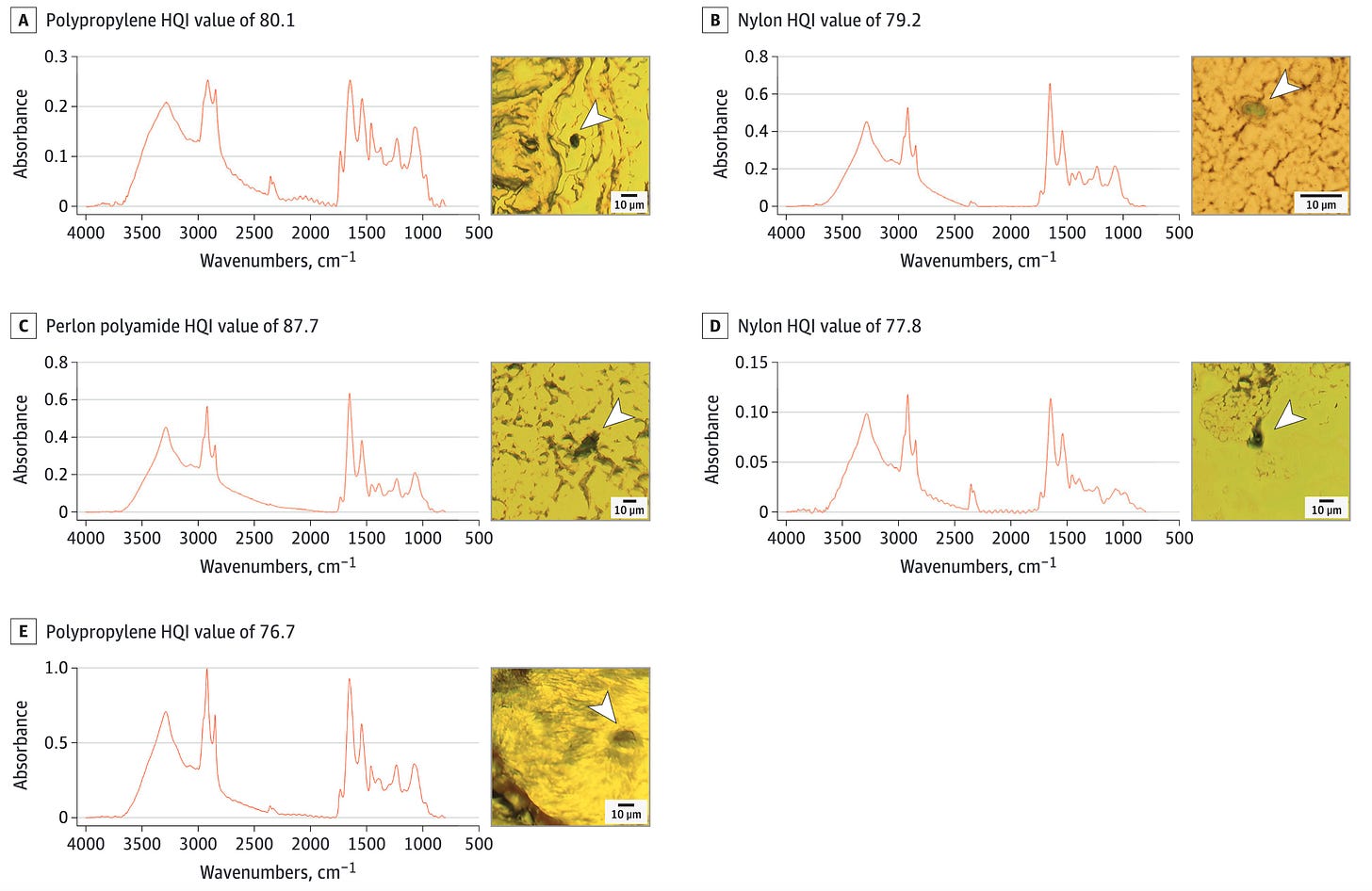
Every so often I come across an article indicating that high amounts of plastic contaminants can be found in our food and drinking water. This week, I read another such disturbing article, published in JAMA Network Open, which has uncovered the presence of microplastics (MPs) in the human olfactory bulb (region of the brain processing smell). This article further claims to provide the first direct evidence that these tiny plastic particles can reach brain tissues through this route (Armato-Lourenco et al., 2024). The study, conducted in São Paulo, Brazil, involved the analysis of olfactory bulb tissues from 15 deceased individuals. Researchers used advanced spectroscopy techniques to identify and characterize the MPs in these tissues, revealing a variety of synthetic polymer particles and fibers, with polypropylene being the most common polymer detected.
The study identified MPs in 8 out of the 15 samples, with particles ranging from 5.5 to 26.4 micrometers in size (check out the figure above). These MPs likely entered the brain through the olfactory pathway, bypassing the blood-brain barrier via the nasal passage and reaching the olfactory bulb through the cribriform plate. This pathway has long been considered a potential route for environmental contaminants to access the brain. Importantly, the study underscores the potential for MPs to cause neurotoxic effects, raising concerns about the health implications of long-term exposure to airborne microplastics.
The discovery that microplastics can reach the brain opens new questions about their impact on human health, particularly in relation to neurological disorders. As the ubiquity of microplastic pollution continues to grow, this study emphasizes the need for further research into the possible effects of MPs on brain function and the risks posed by chronic exposure to these contaminants.
Support:
These newsletters take a significant amount of effort to put together and are totally for the benefit of the reader. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Subscribed
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
Amato-Lourenço, L.F., Dantas, K.C., Júnior, G.R., Paes, V.R., Ando, R.A., de Oliveira Freitas, R., da Costa, O.M.M.M., Rabelo, R.S., Bispo, K.C.S., Carvalho-Oliveira, R. and Mauad, T., 2024. Microplastics in the Olfactory Bulb of the Human Brain. JAMA Network Open, 7(9), pp.e2440018-e2440018.
Iyer, D.P., Khoei, H.H., van der Weijden, V.A., Kagawa, H., Pradhan, S.J., Novatchkova, M., McCarthy, A., Rayon, T., Simon, C.S., Dunkel, I. and Wamaitha, S.E., 2024. mTOR activity paces human blastocyst stage developmental progression. Cell.
Lin, A., Yang, R., Dorkenwald, S. et al. Network statistics of the whole-brain connectome of Drosophila. Nature 634, 153–165 (2024
Mannick, J.B. and Lamming, D.W., 2023. Targeting the biology of aging with mTOR inhibitors. Nature Aging, 3(6), pp.642-660.
Wang, S., Du, Y., Zhang, B., Meng, G., Liu, Z., Liew, S.Y., Liang, R., Zhang, Z., Cai, X., Wu, S. and Gao, W., 2024. Transplantation of chemically induced pluripotent stem-cell-derived islets under abdominal anterior rectus sheath in a type 1 diabetes patient. Cell.
Wu, J., Li, T., Guo, M., Ji, J., Meng, X., Fu, T., Nie, T., Wei, T., Zhou, Y., Dong, W. and Zhang, M., 2024. Treating a type 2 diabetic patient with impaired pancreatic islet function by personalized endoderm stem cell-derived islet tissue. Cell Discovery, 10(1), p.45.


Fantastic round-up, David. I'd read about the fruit fly brain map but not the islets insulin study. Incredible.
Was there a reason for the placement of the islets into the person? Do they migrate anywhere once introduced? Quite impressive if they can function away from the pancreas, although I suppose there's no reason for them not to provided they can still sense blood glucose levels and respond accordingly.
What a mix of fascinating discoveries - some good and some horrifying.