Happy Monday Morning, Readers. Let’s be relentless this week!
In this week’s BioWire, we explore fascinating topics that highlight advancements in biotechnology, including the commercialization of animal cloning, new drugs that enhance muscle growth in clinical studies, the potential of repurposing GLP-1 drugs for dementia, the rise of an academic black market for citation farming, and the ongoing turbulence in the biopharma industry.
As always, if you find these articles valuable, consider supporting by liking, commenting, or sharing with a friend. Let’s get started.
Pet Cloning is Now a Commercialized Market

Have you ever wondered about pet cloning? Admittedly, I hadn’t either, but I stumbled upon Viagen, a company that has made this entire process available to consumers! Viagen is a leader in the pet cloning industry, offering pet owners the opportunity to clone their existing or potentially even deceased animals.
How does this work? The process begins with a biopsy, where a small tissue sample is taken from the pet and sent to Viagen's laboratory. This sample is then used to create a clone through somatic cell nuclear transfer, a sophisticated technique where the nucleus of the donor cell is transferred into an egg cell that has had its own nucleus removed. The resulting embryo is then implanted into a surrogate mother, leading to the birth of a clone genetically identical to the original pet.
But what does this cost? Cloning a pet with Viagen doesn’t come cheap, with prices starting at around $50,000 for dogs and $35,000 for cats. The company also offers cloning services for horses, which has attracted breeders and competitive animal owners, with costs reaching upwards of $85,000. Beyond domestic pets, Viagen has ventured into cloning endangered species, contributing to conservation efforts by preserving the genetic diversity of at-risk animals.
While the price may be steep, a dedicated customer base has emerged, willing to invest in cloning to maintain a connection with their animals or preserve valuable genetic lines. As biotechnology continues to advance, Viagen’s services offer a glimpse into the future of pet ownership. Rather than getting into the ethical questions regarding cloning, I’ll end with a question to you: would you clone your pet?
Targeting Myostatin and Activin A for Muscle Growth and Fat Reduction
The myostatin pathway has long been known as the body’s natural brake on muscle growth. Without this pathway, muscles can grow uncontrollably, as seen in animals with mutations in the myostatin gene. This makes the pathway an appealing target for therapies aimed at increasing muscle mass, particularly for those with sarcopenia or other muscle-related diseases. By blocking this pathway, we can potentially unleash significant muscle development. The pathway itself consists of myostatin, the signaling molecule, and activin, the receptor. When activated, it signals for a reduction in muscle growth and even the breakdown of existing muscle tissue. Both myostatin and the activin receptor present unique opportunities for therapeutic intervention.
In a recent Phase 1 trial, researchers tested the effects of blocking both myostatin and activin using monoclonal antibodies—trevogrumab and garetosmab (Trotter et al, 2024). The study, which included healthy male and postmenopausal female participants, found that the combination therapy significantly increased thigh muscle volume and lean mass while reducing fat mass. These effects were not only dose-dependent but also greater than what could be achieved by targeting myostatin or activin A alone.
This dual blockade could represent a powerful approach to muscle enhancement and fat reduction, particularly for conditions where muscle preservation is key. While this was only a Phase 1 trial, the involvement of Regeneron Pharmaceuticals, a major player in the biotech industry, signals the potential significance of these findings. Regeneron’s backing highlights the company’s interest in staying competitive in the expanding weight loss market, and their support suggests that this research could lead to new and impactful therapeutic strategies.
GLP-1 Receptor Agonists in Alzheimer’s Disease
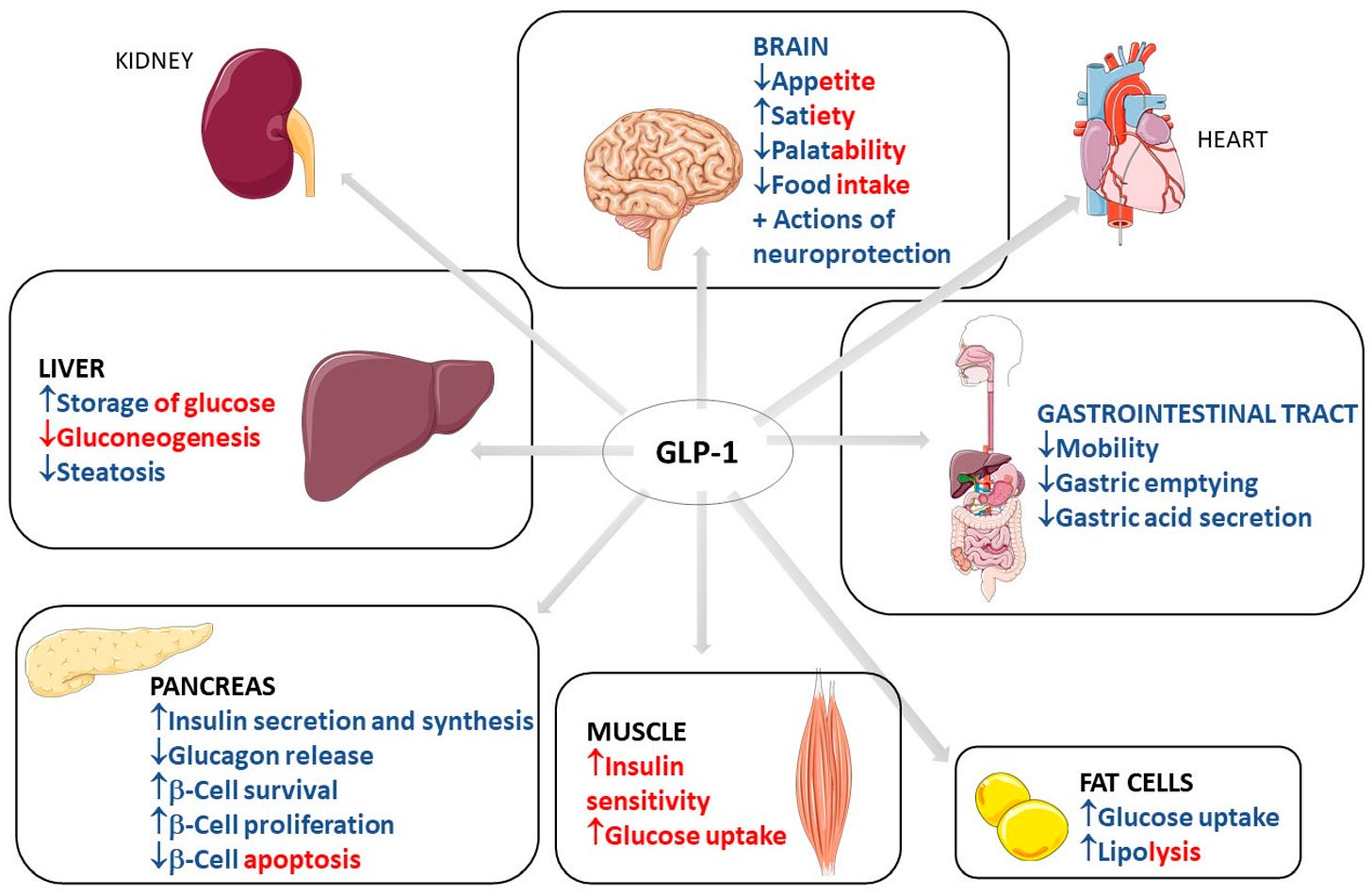
It seems like every week we’re talking about Glucagon-like peptide 1 receptor agonists (GLP-1) drugs, like semaglutide or tirzepatide, for their antidiabetic effects. Now, researchers are exploring their potential role in neurodegenerative diseases like Alzheimer’s. To be blunt, it’s unclear if this indicates a link between Alzheimer’s and metabolism, showcases the power of GLP-1 drugs and their vast applications, or reveals our desperation for effective Alzheimer’s therapies after decades of underwhelming research and drug development.
Regardless, a recent systematic review explored whether GLP-1 drugs could impact core Alzheimer’s disease pathology, such as amyloid-β (Aβ) plaques and tau neurofibrillary tangles, and improve cognitive function (Liang et al, 2024). Unfortunately, the findings were underwhelming—GLP-1 therapies did not significantly alter Aβ or tau biomarkers, nor did they improve cognitive outcomes in clinical trials.
Okay, so GLP-1 drugs seem fairly ineffective as a therapeutic against Alzheimer’s disease symptoms. To be fair, this was always a long shot. However, there’s a silver lining. The review highlighted potential metabolic benefits, such as improved glucose metabolism and a possible increase in cerebral glucose transport, suggesting neuroprotective effects. While GLP-1 drugs may not directly impact the primary hallmarks of Alzheimer’s, they could still play a supportive role in managing metabolic dysfunction. Further large-scale studies are needed to determine whether these metabolic improvements translate into meaningful clinical outcomes for patients with Alzheimer’s disease.
Academia’s Citation Black Market
In academia, the success of professors and students alike is mostly measured by the number of publications they have and the impact those publications generate. It’s no surprise that this is referred to as the currency of academia. Citation counts on platforms like Google Scholar and PubMed are commonly used to track this impact. These citations are linked to researchers’ profiles, influencing their status, career advancement, promotions, and ability to secure grants. With so much at stake, the integrity of these metrics is crucial—or perhaps just another facet of the increasingly toxic academic environment.
In related news, a troubling (or somewhat predictable) new trend is emerging in the academic world: the rise of a citation black market. This involves the buying and selling of fake or manipulated citations to artificially boost researchers' citation counts—a practice with serious implications for academic integrity. A recent sting operation revealed that citations could be purchased in bulk, with 50 citations available for just $300. These citations were added to a fake Google Scholar profile, showcasing how easily these metrics can be manipulated.
So far, the response from academic institutions and platforms like Google Scholar has been relatively muted. This is not surprising considering Google Scholar remains nearly identical to its state in 2010 (when I first started using it)— showcasing Google’s disinterest in the platform. While there are some efforts to detect and prevent citation manipulation, the rapidly evolving nature of these schemes poses a significant challenge. It’s crucial for the academic community to confront the toxic culture that has emerged in universities, where the h-index seems to overshadow meaningful scientific inquiry.
Biopharma remains in choppy waters, but there is light at the end of the tunnel
The biopharma industry has faced significant layoffs in recent months, with companies like Biogen, Amgen, and others making difficult decisions to reduce their workforce. These job cuts are not isolated incidents but appear to be part of a broader trend of economic recalibration. The U.S. Bureau of Labor Statistics (BLS) has indicated that it will be revising job numbers downward by up to 1 million for the period from April 2023 to March 2024, revealing that the labor market may have been significantly weaker than initially reported. This substantial correction has raised concerns about the stability of various sectors, including biopharma, which is known for its high R&D costs and reliance on investor confidence.
The layoffs in biopharma have primarily affected roles in research and development (R&D), manufacturing, and sales. Companies are scaling back on early-stage R&D projects due to the high costs and uncertainties associated with drug development. Additionally, with the rise of remote healthcare and digital solutions, traditional sales roles are being impacted as companies shift towards more technology-driven strategies. These layoffs reflect the industry's need to streamline operations, reduce costs, and focus on core projects in a challenging financial environment.
However, it's not all gloom and doom for the biopharma industry. In the second quarter of 2024, venture capital (VC) funding in biopharma surged to $9.2 billion across 215 deals, the highest level since the same quarter in 2022. This increase from the $7.4 billion reported in the previous quarter highlights renewed confidence in the sector. The funding boost is driven in part by large investments in AI and obesity-related biotech, though the exit landscape remains challenging, with slower M&A activity and fewer IPOs. Despite these hurdles, the continued influx of VC funding suggests that investors believe in the long-term potential of biopharma innovation, particularly in areas like precision medicine and digital health.
Support:
These newsletters take a significant amount of effort to put together and are totally for the benefit of the reader. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
https://www.viagenpets.com/?utm_source=nxst&utm_medium=google&utm_campaign=fall2023&gad_source=1&gclid=Cj0KCQjwrKu2BhDkARIsAD7GBotUV5SBJOzEyQQxBbuYVweapZMA7xUE2nOR8vOZiavWfTqboBrkaLoaAqmPEALw_wcB
https://www.nature.com/articles/d41586-024-01672-7
https://www.forbes.com/sites/dereksaul/2024/08/21/us-added-818000-fewer-jobs-than-previously-thought-from-march-2023-to-march-2024-government-says/
https://www.fiercebiotech.com./biotech/biopharma-vc-hits-highest-level-2022-challenges-exit-landscape-remain
GONZALEZ TROTTER, D.I.N.K.O., DONAHUE, S., WYNNE, C., ALI, S., PARASOGLOU, P., BOYAPATI, A., MOHAMMADI, K., MUSSER, B.J., MEIER, P., MASTAITIS, J. and GASPARINO, E., 2024. 34-OR: The Effect of Combined Activin A and Myostatin Blockade on Body Composition—A Phase 1 Trial. Diabetes, 73(Supplement_1).
Laurindo, L.F., Barbalho, S.M., Guiguer, E.L., da Silva Soares de Souza, M., de Souza, G.A., Fidalgo, T.M., Araújo, A.C., de Souza Gonzaga, H.F., de Bortoli Teixeira, D., de Oliveira Silva Ullmann, T. and Sloan, K.P., 2022. GLP-1a: going beyond traditional use. International Journal of Molecular Sciences, 23(2), p.739.
Liang, Y., Doré, V., Rowe, C.C. and Krishnadas, N., 2024. Clinical Evidence for GLP-1 Receptor Agonists in Alzheimer’s Disease: A Systematic Review. Journal of Alzheimer's Disease Reports, 8(1), pp.777-789.




I love the idea of cloning, but as I understand it, it's not remotely like getting a second copy of the pet you love. Being genetically a twin is one thing. Being the second coming is something else. It sounds like cloning would have a much more practical application with livestock, assuming the breed registry allows it. Thoroughbreds have to be produced by a live cover to be registered in the studbook. I'm pretty sure that's not the case with Standardbreds, and I know it's not the case with sport horses, which can be produced with AI. They'd be good candidates for cloning, since the aim would be furthering the genetic profile and not recreating a specific pet's personality. Same for cattle. Several of my cows are bred by AI (which is unbelievably cheap) because it gives me access to bulls that I don't have to keep on my own property. Cloning would have to come way down in price to be competitive with AI for livestock producers, but it makes sense if one of the parents of the clonee isn't available any more and if there's no frozen semen available.
It's funny (not ha-ha funny) that the same people who leap to label skeptics and critics as "science deniers" on everything from "covid" to climate change, and who claim to deplore mis/dis/mal-information, are some of the same people who have absolutely nothing to say about the fraud perpetrated by "researchers" who buy citations. Is it a massive scandal that everyone's talking about? Has it galvanized insiders into insisting on integrity and honesty? I read a lot, but I haven't heard anything about it.
Medical errors are the third-leading cause of death in the US. Every year. Year after year. If the airlines crashed a fully-loaded 767 and 747 three days in a row, no one would get on an airplane again. An equivalent number die daily from medical errors. When 2-3 airliners crashed every year, the industry understood that something had to change, and something did. It took fifty years of hard work and hard lessons, but it happened. Medicine? Researchers in laboratory and academic settings? Meh. Researchers aren't crashing airplanes or mis-dosing patients, but I don't understand the indifference or even apparent hostility toward one's own career, integrity, and profession.
What an interesting summary about citation fraud. While Google scholar is a great resource, I don't think Google qualifies as a metric for academics research standing, particularly, if they don't have in place reliable measures to validate the metrics.