Happy Monday Morning, Readers. As always, let’s be relentless!
This week’s edition of BioWire will cover a number of interesting topics at the interface of biomedicine, biotechnology, and computational sciences:
The world's first human trial for a drug that regenerates teeth gives the potential for a permanent solution for tooth loss.
Soterios Pharma's Phase 2 success with a topical alopecia treatment that addresses inflammatory and autoimmune aspects of hair loss.
The impressive results of CD19 CAR-T cell therapy in treating severe autoimmune diseases show potential for long-term remission.
Johnson & Johnson's pivotal trial of seltorexant, a new treatment significantly improving depressive symptoms and sleep disturbances in MDD patients with insomnia by targeting the orexin-2 receptor.
FinalSpark's Neuroplatform is a bioprocessor utilizing human brain organoids for biocomputing.
How will these breakthroughs shape the future of medicine and technology?
First Human Trials Set to Begin for Tooth Regrowth Drug
The world's first human trial for a drug capable of regenerating teeth is set to begin at Kyoto University Hospital from September 2024 to August 2025, following successful animal trials. This pioneering treatment aims to help people suffering from tooth loss by deactivating the USAG-1 protein, which suppresses tooth growth, thereby encouraging bone morphogenetic protein (BMP) signaling to generate new teeth (Murashima-Suginami et al, 2021). The trial will initially involve 30 males aged 30-64 missing at least one molar, with plans to expand to children aged 2-7 with congenital tooth deficiency and eventually to individuals with partial edentulism. If successful, the drug could be commercially available by 2030, offering a permanent solution for tooth loss (New Atlas).
Hair Loss Drug Shows Promise with Phase 2 Success in Alopecia Trial
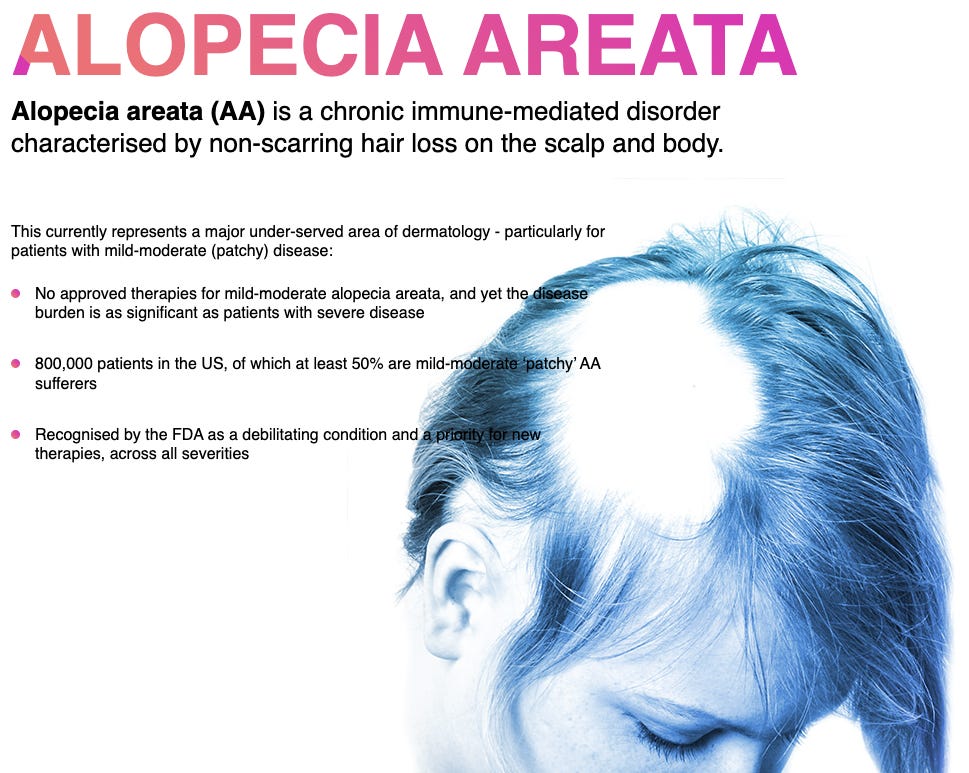
Soterios Pharma's alopecia areata cream, STS-01, has achieved a successful Phase 2 trial, showing a significant improvement in hair regrowth for patients with mild to moderate alopecia areata. The 1% dose cohort saw a 76% improvement in severity, with 18% experiencing total hair regrowth. This non-steroidal topical therapy works by modulating inflammatory responses and T-cell proliferation, offering an alternative to JAK inhibitors like Eli Lilly's Olumiant. Soterios plans to advance to Phase 3 trials and aims to become the first approved therapy for mild/moderate alopecia areata. Read the full press release here.
CAR-T Cell Therapy Achieves Remarkable Results in Autoimmune Disorders
Researchers at University Hospital Erlangen in Germany have demonstrated the potential of CD19 CAR-T cell therapy to treat severe cases of systemic lupus erythematosus (SLE). In a study published in the New England Journal of Medicine, 15 patients with severe autoimmune conditions, including SLE, idiopathic inflammatory myositis, and systemic sclerosis, received a single infusion of CD19 CAR-T cells after preconditioning with fludarabine and cyclophosphamide.
The results were remarkable: all patients with SLE achieved remission according to DORIS criteria, and significant clinical improvements were observed in patients with the other conditions. This study highlights the potential of CAR-T cell therapy to induce long-term remission in autoimmune diseases by targeting and eliminating autoreactive B cells. Notably, the treatment allowed patients to discontinue other immunosuppressive therapies, with manageable side effects including mild cytokine release syndrome (Muller et al, 2024).
Seltorexant Phase 3 Trial Shows Significant Improvement in Depression and Sleep for MDD Patients
Johnson & Johnson's pivotal Phase 3 trial of seltorexant demonstrated significant improvements in depressive symptoms and sleep disturbances in patients with major depressive disorder (MDD) who suffer from insomnia. Unlike traditional antidepressants that target serotonin and norepinephrine pathways, seltorexant works by selectively antagonizing the orexin-2 receptor, which regulates wakefulness and arousal. This dual-action mechanism helps improve both mood and sleep quality, making it a promising treatment for patients who have not responded well to conventional therapies. The drug was well-tolerated, with a safety profile consistent with previous trials. Read the full press release here.
FinalSpark Develops a Platform for Wetware Computing

The Swiss startup, FinalSpark, has introduced Neuroplatform, the the world's first bioprocessor using human brain organoids (Jordan et al, 2024). Such wetware computing using living intelligent organoid systems is an emerging field at the intersection of electrophysiology and artificial intelligence. This platform uses living neurons to perform computations. However, unlike Artificial Neural Networks (ANNs), which update digital tensors to modify network responses, this field requires new methods for biological neural networks. The Neuroplatform was developed to facilitate these methods and allows for large-scale electrophysiological experiments on neural organoids with lifespans exceeding 100 days. It features a streamlined process for organoid production, 24/7 action potential monitoring, electrical stimulation, and an automated microfluidic system to ensure stable conditions. Over three years, it has been used with over 1,000 organoids, collecting more than 18 terabytes of data. Researchers can remotely conduct experiments through a dedicated API, supporting complex operations and real-time adjustments, and the platform is freely available for research purposes. Learn more here.
A Virtual Hospital Ran by ChatGPT!
Support:
These newsletters take a significant amount of effort to put together and are totally for the benefit of the reader. If you find these explorations valuable, there are multiple ways to show your support:
Engage: Like or comment on posts to join the conversation.
Subscribe: Never miss an update by subscribing to the Substack.
Share: Help spread the word by sharing posts with friends directly or on social media.
References:
Jordan, F.D., Kutter, M., Comby, J.M., Brozzi, F. and Kurtys, E., 2024. Open and remotely accessible Neuroplatform for research in wetware computing. Frontiers in Artificial Intelligence, 7, p.1376042.
Murashima-Suginami, A., Kiso, H., Tokita, Y., Mihara, E., Nambu, Y., Uozumi, R., Tabata, Y., Bessho, K., Takagi, J., Sugai, M. and Takahashi, K., 2021. Anti–USAG-1 therapy for tooth regeneration through enhanced BMP signaling. Science advances, 7(7), p.eabf1798.
Müller, F., Taubmann, J., Bucci, L., Wilhelm, A., Bergmann, C., Völkl, S., Aigner, M., Rothe, T., Minopoulou, I., Tur, C. and Knitza, J., 2024. CD19 CAR T-Cell Therapy in Autoimmune Disease—A Case Series with Follow-up. New England Journal of Medicine, 390(8), pp.687-700.
https://newatlas.com/medical/tooth-regrowing-human-trial/
https://www.prnewswire.com/news-releases/soterios-pharma-announces-positive-topline-results-from-phase-ii-study-of-sts-01-in-the-treatment-of-mild--moderate-alopecia-areata-302157485.html
https://www.jnj.com/media-center/press-releases/johnson-johnson-pivotal-study-of-seltorexant-shows-statistically-significant-and-clinically-meaningful-improvement-in-depressive-symptoms-and-sleep-disturbance-outcomes



I wonder what virtual healthcare is like